Computational Modeling:
A Bridge to Precision Medicine
Our lab uses mathematical modeling to unify neural physiology, device physics, and clinical outcomes. As a newly launched lab, we build on our PI’s decades of prior research and translational development in closed-loop neuromodulation. We develop mechanistic and data-driven computational models of stimulation delivery and brain/spinal network response to predict treatment effects and optimize patient-specific neuromodulation for safety and efficacy.
1. Modeling Neural Pathway Dynamics
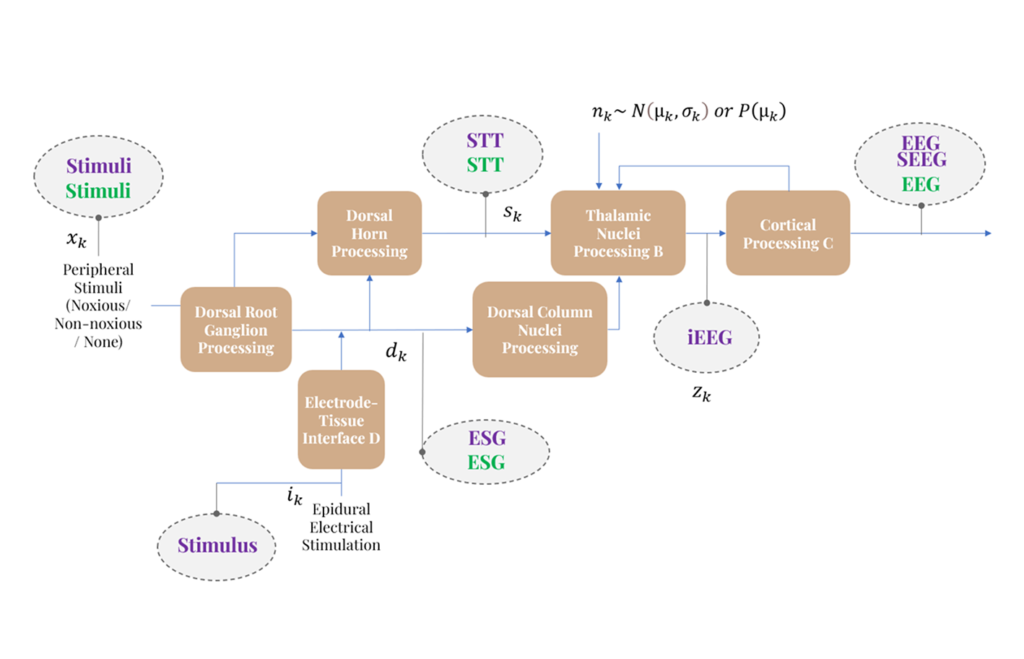
We develop models to track how information propagates through the body to the brain.
We characterize the flow of signals from a stimulus (e.g., touch or pain) to the cortex.
Using system identification and state estimation, our models estimate internal dynamics and latent state variables that govern responses across inputs and conditions.
Currently being worked on
2. Smart Model-Predicted Personalized Dosing
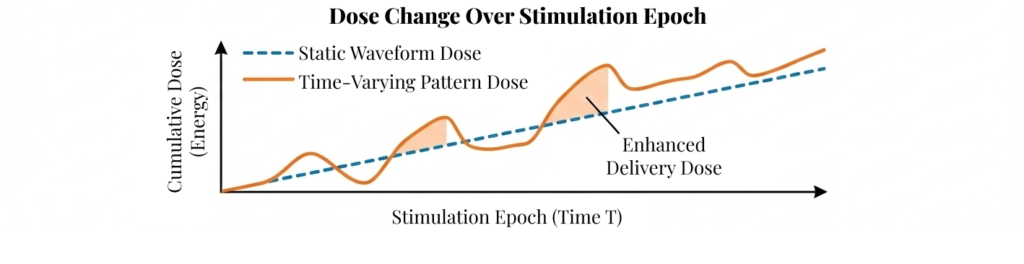
Traditional neuromodulation often relies on "static" waveforms (fixed amplitude, frequency, pulse width). We are developing time-varying pulse-pattern stimulation that adapts therapy “dose” over time.
Our models adjust stimulation parameters to shape the delivered “dose rate” across time, matching the patient’s changing needs.
We track dose metrics to keep therapy effective while staying within predefined safe limits and minimizing side effects.
3. Adaptive Stimulation Therapy
We cast neuromodulation in a systems-and-control framework, where sensing and stimulation form a closed feedback loop across the brain and spinal cord.
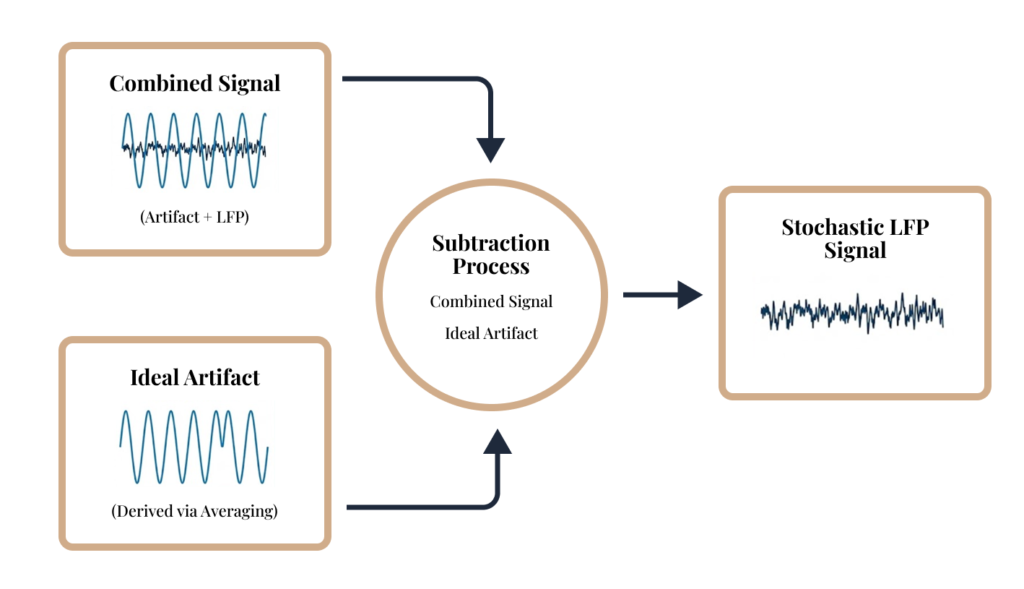
We model and suppress stimulation artifacts to recover interpretable neurophysiological signals in real-time.
We translate individual neural dynamics into control-relevant features and therapy targets.
Using state estimation and biomarker-driven decision logic, therapy is adjusted to suppress pathological rhythms and restore network balance. Recent work we presented at AES (Dec 2025) identified seizure-termination network markers (e.g., PLV) that may reflect therapy-driven plasticity and could inform adaptive control policies.