Unifying Approach
Across Our Four Pillars
We build biomarker-driven, safety -constrained closed-loop neuromodulation by integrating multimodal sensing, computational modeling, control policy design, and IRB-based translational studies.
This pillar provides the biomarker and state-estimation layer that informs modeling and closed-loop control pillars.
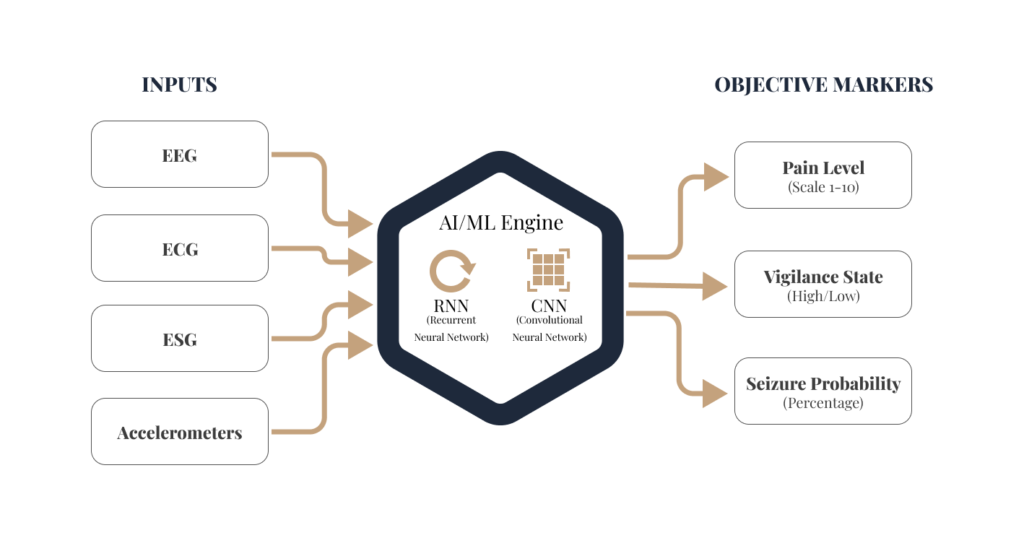
Besides advanced signal processing techniques, our lab leverages cutting-edge Artificial Intelligence (AI) and Machine Learning (ML) to transform subjective patient experiences into objective, measurable biomarkers. By integrating multimodal data from neural and physiological sources, we develop predictive models that define “neural signatures” for complex medical conditions (e.g. drug-resistant epilepsy and chronic pain).
Traditional diagnostics often rely on intermittent snapshots of a patient’s state. Our approach utilizes continuous, multimodal sensing to create a comprehensive patient’s health profile.
We employ a advanced algorithmic frameworks (AI and ML architectures) tailored to the specific dynamics of the human nervous system investigated.
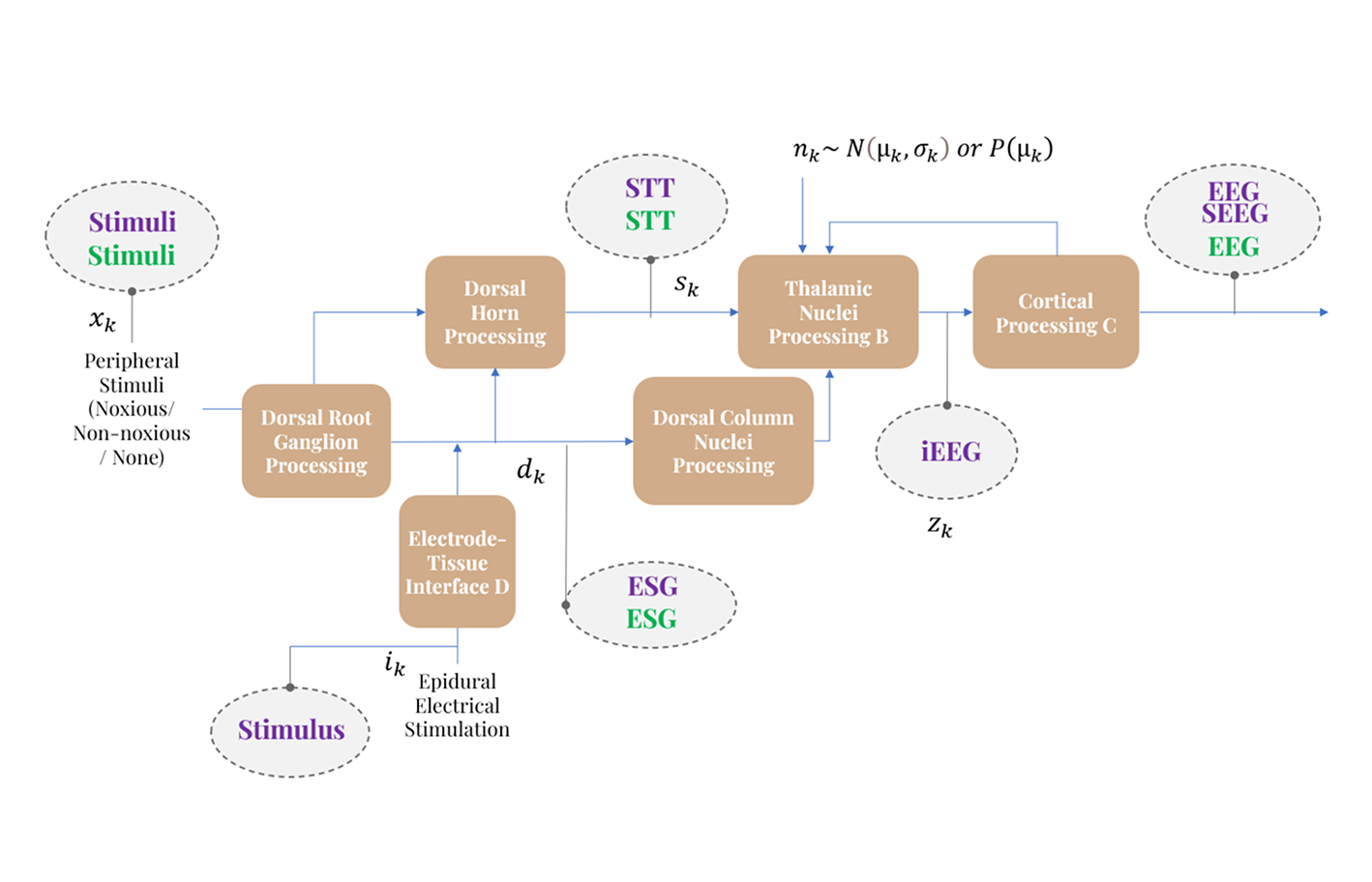
Our lab uses computational modeling to connect neural physiology, device physics, and clinical outcomes. We develop mechanistic and data-driven models across scales, from brain/spinal network dynamics (state-space, system identification, and control frameworks) to neural-circuits biophysics to characterize neurophysiological events and to design stimulation strategies that adapt to measured network. The goal is to predict treatment effects and enable patient-specific neuromodulation that is optimized for safety and efficacy.
Modeling Neural Pathway Dynamics
We develop models to track how information propagates through the body to the brain.
- Pathway Mapping – We characterize the flow of signals from a stimulus (e.g., touch or pain) to the cortex.
- Predicting Internal State – Using system identification and state estimation, our models estimate internal dynamics and latent state variables that govern responses across inputs and conditions.
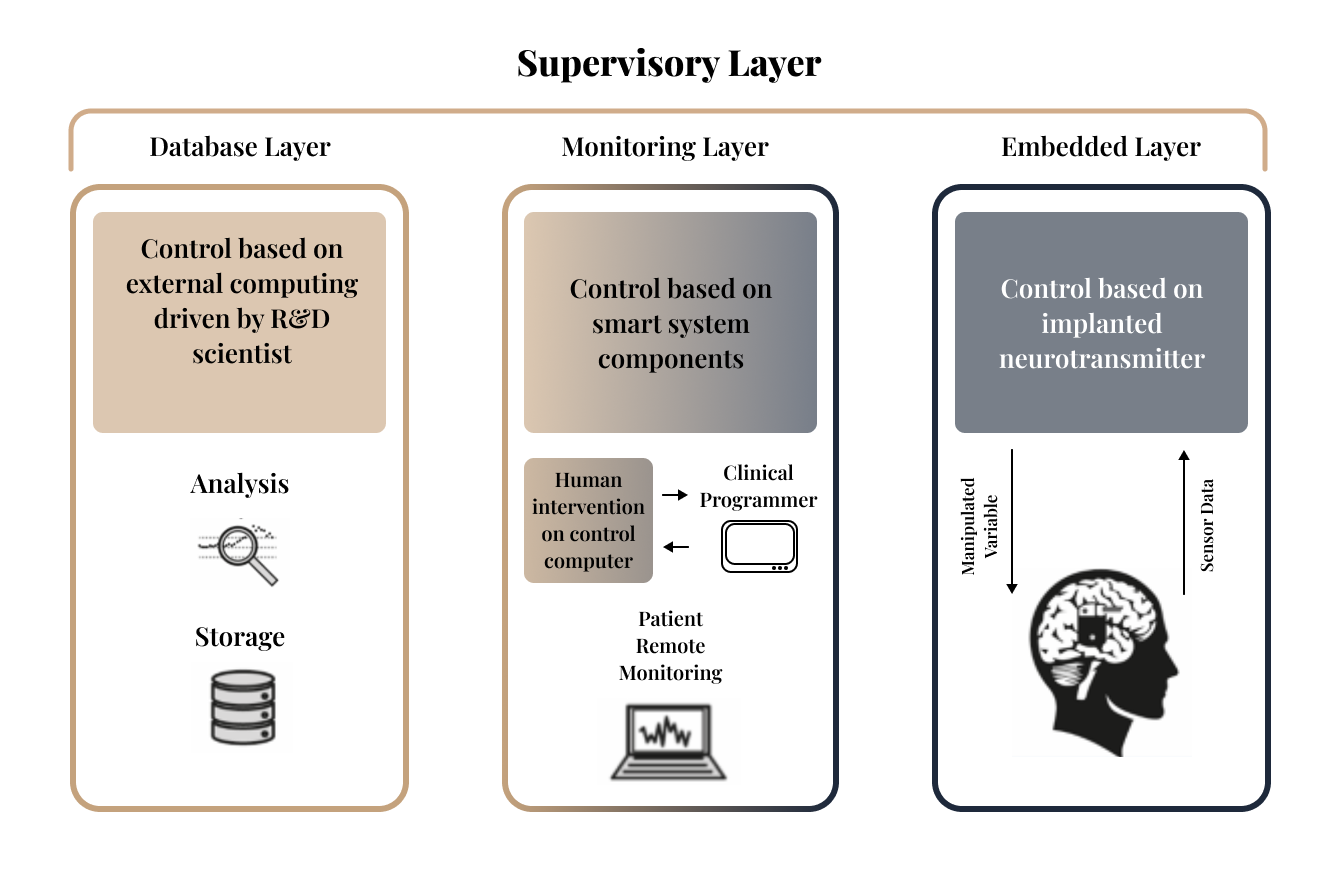
A closed-loop system is an intelligent feedback loop that monitors a patient’s biological state and automatically adjusts therapy to maintain (or drive toward) a desired outcome. Our lab develops and studies closed-loop neuromodulation through an integrated three-layer architecture described below. While individual projects may emphasize one layer, embedded control, clinical monitoring, or data-driven refinement, they share a common foundation in biomarker/state definitions, safety constraints, and outcome evaluation, ensuring results translate cleanly across the full pipeline.
Embedded Layer
This is the patient-side interface. An implanted or externalized neurostimulator acquires biosignals, performs on-device processing (as appropriate), and adjusts therapeutic parameters. Control action can occur across multiple updates time scales, from real time to hours, days, and longer horizons, to support both rapid physiological regulation and longer-term personalization.
Monitoring Layer
This layer enables supervised care and remote oversight. Clinicians can review signals and therapy history, update programming and safety limits, and intervene when needed. In parallel, connected tools support secure remote monitoring so therapy can be evaluated and adjusted without requiring frequent in-person visits.
Database Layer
This is our research hub. We managed longitudinal data, run deeper analysis, and refine models an control policies, so algorithms improve over time and can be translated into safer, more effective closed-loop therapies.
Together, these layers implement our end-to-end pipeline: sensing, biomarker and state estimation, model-informed decision logic, adaptive stimulation, and outcome evaluation.
We run IRB-approved protocols designed to maximize information per participant and accelerate iteration of neuromodulation parameters and biomarkers. Each study visit is treated as a controlled experiment: we deliver structured stimulation paradigms, capture synchronized measurements, and rapidly translate findings into improved personalization and closed-loop design.
“Smart/Optimized” reflects three design principles: standardized clinical endpoints with low-burden digital capture (e.g., ePRO/eDiary) alongside clinician- and objective measures; structured multimodal sensing during controlled stimulation; analysis pipelines that enable rapid phenotype/state characterization and patient-specific dose–response mapping.
Our current emphasis is multimodal data collection in-clinic. While our current emphasis is in-clinic multimodal studies, the same measurement and analysis principles are designed to extend to longitudinal, real-world settings
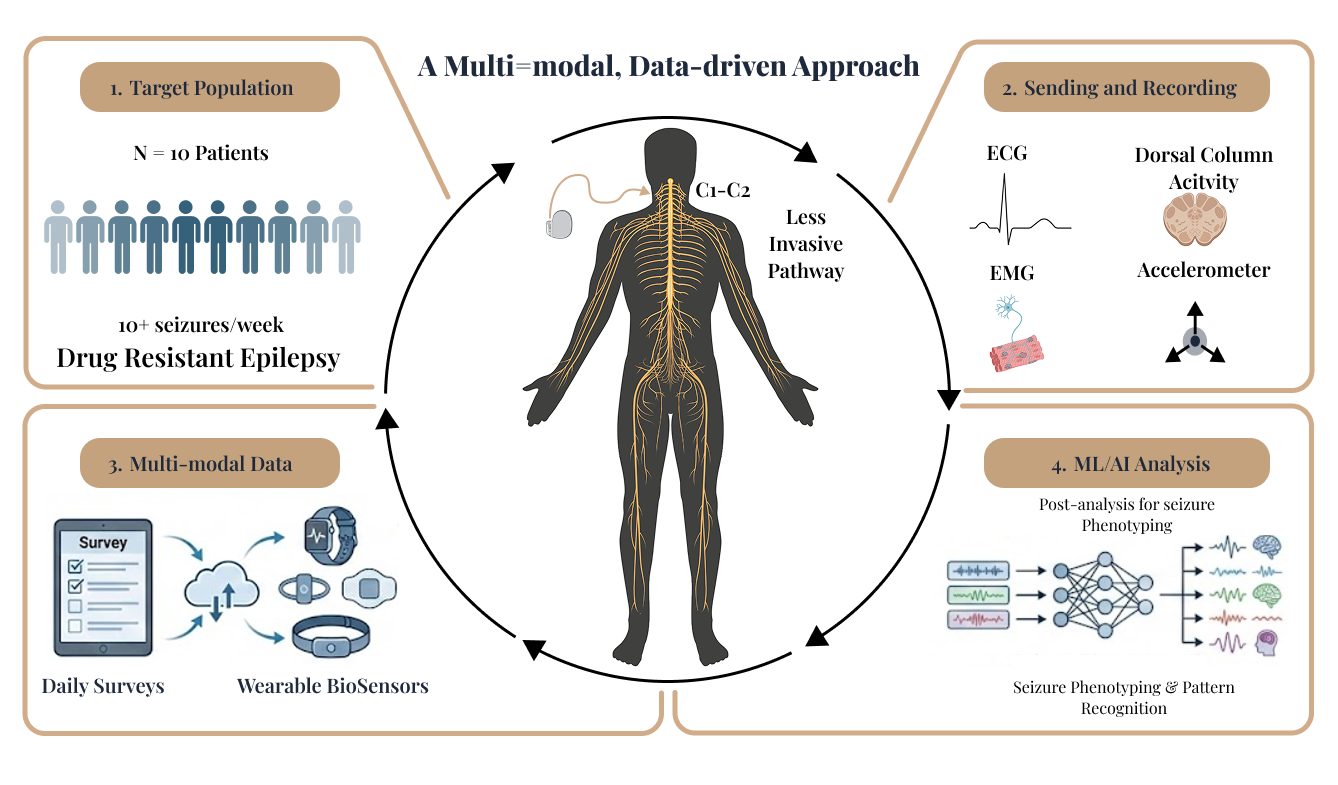
A Multimodal, Data-Driven Approach
We are initiating in-clinic studies that pair controlled stimulation with synchronized neural and physiological recordings to quantify acute response, identify candidate biomarkers, and inform subsequent personalization across neurological conditions.
This translational engine supports all pillars by enabling consistent measurement, rapid iteration, and clinically grounded validation.