Redefining Research:
Smart & Optimized Clinical Trials
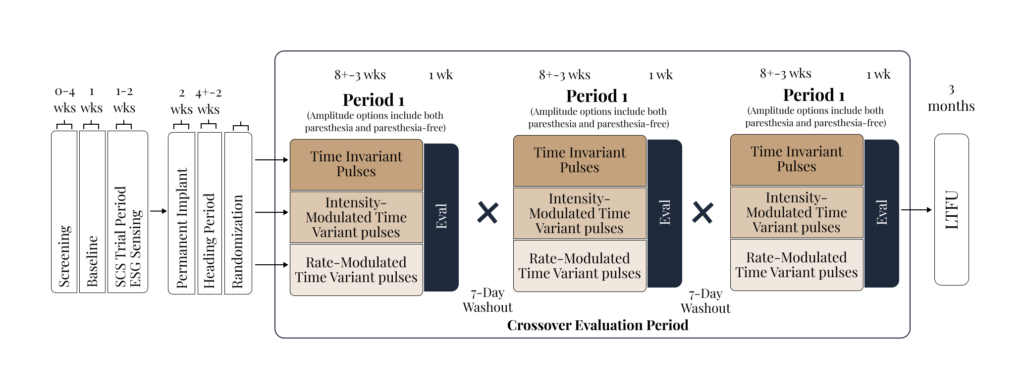
We run IRB-based study protocols designed to maximize information per participant and accelerate iteration of neuromodulation parameters and biomarkers. “Smart/Optimized” refers to: (i) standardized clinical endpoints with digital capture (e.g., ePRO/eDiary) alongside objective measures, and (iii) analysis pipelines that support rapid phenotype/state characterization and patient-specific dose–response mapping.
What It Is: A Multimodal, Data-driven Approach
We are initiating in-clinic studies that pair controlled stimulation with synchronized neural + physiological recordings to quantify acute response, identify candidate biomarkers, and inform subsequent personalization and closed-loop design across neurological conditions.
Initial studies include healthy volunteers and participants with neurological conditions with a planned expansion to epilepsy/DRE cohorts and chronic pain this year.
We integrate high-resolution neural and physiological signals to quantify state and acute response.
Our studies are designed for multimodal data collection in-clinic. Real-world monitoring is coming.
Data is analyzed using advanced signal processing and ML/AI algorithm to support phenotyping, state tracking, and patient-specific signatures that inform study design and therapy optimization.
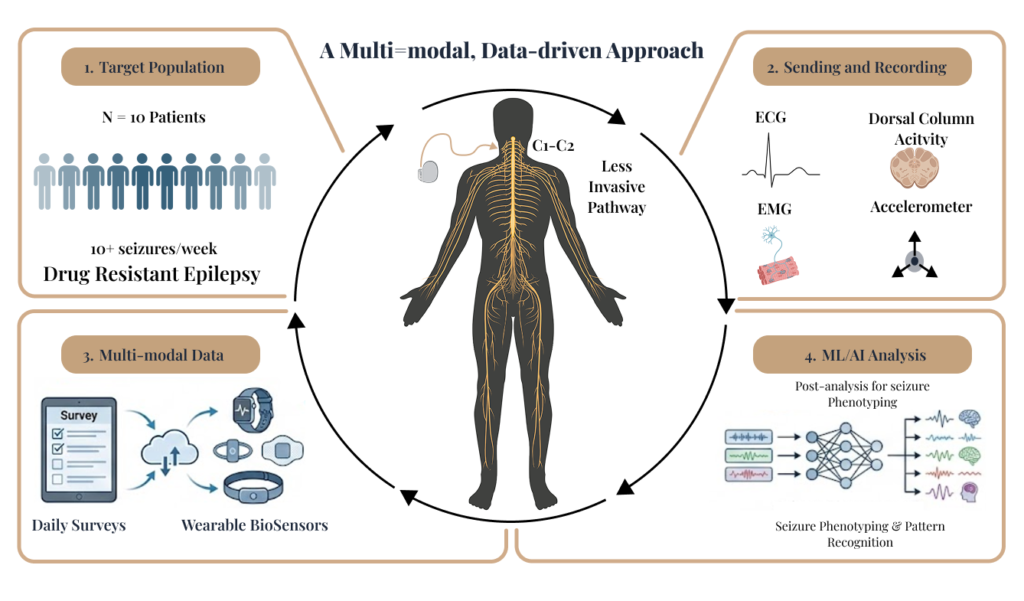
What We Do: Targeting Less Invasive Pathways
Our research seeks to provide relief with less risk. We are initiating feasibility studies to evaluate novel, less invasive neural pathways for neuromodulation therapy.
Our primary research goals include:
Assessing the safety, optimal dosage, and tolerability of different neuromodulation paradigms.
Quantifying how stimulation modulates measurable neurophysiological and autonomic state markers to distinguish clinically relevant states.
Characterizing individual response profiles across a broad parameter space to identify patient-specific settings that perform best.
What We Do: Innovative Dynamic Stimulation Studies